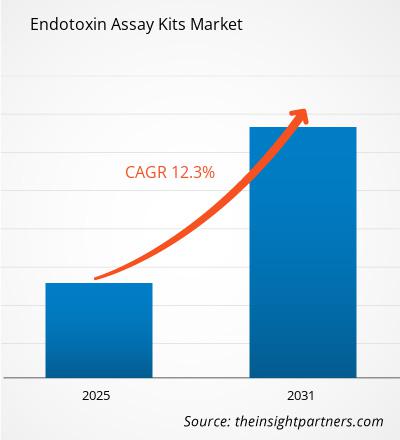
The Endotoxin Assay Kits Market is expected to register a CAGR of 12.3% from 2025 to 2031, with a market size expanding from US$ XX million in 2024 to US$ XX Million by 2031.
The report is segmented by Testing Method (Gel Clot Endotoxin Test, Chromogenic Endotoxin Test, Turbidimetric Endotoxin Test); Product (Endotoxin Detection Products, LAL Test Reagents, Mycoplasma Detection and Removal, PCR Mycoplasma Detection Kit, Mycoplasma Elimination Cocktail, Others); and End User (Biopharmaceutical Companies, Research Institutes, Academic Centers, Others). The global analysis is further broken down at the regional level and major countries. The report offers the value in US$ for the above analysis and segments.
Purpose of the Report
The report Endotoxin Assay Kits Market by The Insight Partners aims to describe the present landscape and future growth, top driving factors, challenges, and opportunities. This will provide insights to various business stakeholders, such as:
- Technology Providers/Manufacturers: To understand the evolving market dynamics and know the potential growth opportunities, enabling them to make informed strategic decisions.
- Investors: To conduct a comprehensive trend analysis regarding the market growth rate, market financial projections, and opportunities that exist across the value chain.
- Regulatory bodies: To regulate policies and police activities in the market with the aim of minimizing abuse, preserving investor trust and confidence, and upholding the integrity and stability of the market.
Endotoxin Assay Kits Market Segmentation
Testing Method
- Gel Clot Endotoxin Test
- Chromogenic Endotoxin Test
- Turbidimetric Endotoxin Test
Product
- Endotoxin Detection Products
- LAL Test Reagents
- Mycoplasma Detection and Removal
- PCR Mycoplasma Detection Kit
- Mycoplasma Elimination Cocktail
End User
- Biopharmaceutical Companies
- Research Institutes
- Academic Centers
Customize This Report To Suit Your Requirement
You will get customization on any report - free of charge - including parts of this report, or country-level analysis, Excel Data pack, as well as avail great offers and discounts for start-ups & universities
Endotoxin Assay Kits Market: Strategic Insights

- Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
Endotoxin Assay Kits Market Growth Drivers
- Increasing Demand for Pharmaceutical and Biopharmaceutical Products: The Endotoxin Assay Kits Market grows because people need more pharmaceutical and biopharmaceutical products. Endotoxins as outer membrane components of specific bacteria create health problems when they appear in pharmaceutical products and medical supplies. The testing of endotoxins now serves as a mandatory quality check during drug production. The pharmaceutical and biopharmaceutical fields worldwide are expanding quickly especially in vaccine production and chronic disease management which drives up demand for endotoxin testing kits. Regulatory bodies including the U.S. FDA and European Medicines Agency require endotoxin testing of drug products vaccines and medical devices to safeguard patient health. Pharmaceutical businesses will need more endotoxin assay kits because they produce more medications and develop personalized medicines in the coming years. Endotoxin assay kit manufacturers find new opportunities to develop better testing solutions because the pharmaceutical and biopharmaceutical market keeps getting bigger. More pharmaceutical clients need endotoxin testing because they work with CROs and CMOs who contract these services.
- Growing Stringent Regulatory Requirements: The Endotoxin Assay Kits Market expands because health authorities worldwide make testing standards stricter in pharmaceutical and medical device sectors. Global health authorities such as FDA and EMA plus other national agencies enforce strict safety standards to test pharmaceuticals and medical products. Regulatory rules require product manufacturers to test for endotoxins because such bacteria could harm patients when they get into these medical items. Healthcare systems work to protect patients so manufacturers need to use very precise and dependable endotoxin testing methods which creates higher demand for endotoxin assay kits. Endotoxin testing now follows established procedures during product development because quality control and production compliance standards keep increasing. The endotoxin assay kit market will grow because future reports show that testing standards must meet industry rules. Businesses manufacturing pharmaceuticals and medical devices that deliver advanced testing solutions to match new standards will take market share from others as healthcare organizations focus more on safety and compliance.
- Advancements in Endotoxin Testing Technology: New technology developments in endotoxin testing help the Endotoxin Assay Kits Market grow. For many years the Limulus Amebocyte Lysate (LAL) assay served as the main method to detect endotoxins in samples. Manufacturers now use advanced biotech methods to build testing equipment that identifies endotoxins faster and with better results. New methods like recombinant factor C (rFC) tests and endotoxin biosensors show better results than traditional LAL-based tests. These improved tests help companies test for endotoxins faster and cheaper with superior detection results. By automating endotoxin testing systems manufacturers now process samples faster and reduce human mistakes. Market analysts expect advanced technology adoption will drive more endotoxin assay kit sales in the future. The market will keep growing because endotoxin assay kits deliver quicker and better results while more companies need high-volume testing for their pharmaceutical and biopharmaceutical work. The market for advanced endotoxin testing solutions will grow because manufacturers who create better testing methods will take more market share from the industry.
Endotoxin Assay Kits Market Future Trends
- Increasing Shift Toward Automated Testing Systems: Companies in the Endotoxin Assay Kits Market are moving to automated test systems for endotoxin detection more often. Automation has changed how healthcare and pharmaceutical companies perform diagnostics and laboratory tests. Endotoxin testing automation lets us test faster while decreasing mistakes and running more tests effectively. These systems handle many endotoxin tests each day which benefits pharmaceutical companies and testing organizations that test frequently. Automated endotoxin testing systems linked to LIMS platforms simplify work processes by showing live results and data records. The pharmaceutical sector and research institutions will require more automated endotoxin testing solutions because they need fast and precise detection methods. Manufacturers will build more popular automated endotoxin testing systems that meet advanced testing requirements. The growth of automation technology companies will gain more market share which creates better prospects for endotoxin assay kit businesses.
- Focus on Multi-Component Assays: Manufacturers in the Endotoxin Assay Kits Market now prioritize developing tests that screen for several contaminants at once including endotoxins. Multi-component assays include various detection methods within one test to check for multiple contaminants in pharmaceutical and biopharmaceutical products at once. This method saves time for testing while producing more results faster and making quality checks better. Manufacturers work to produce testing kits that find endotoxins while tracking other product contaminants like host cell proteins and residual solvents. The medical device industry alongside pharmaceutical and biotechnology sectors will adopt these advanced testing kits because product safety is their top priority. The pharmaceutical industry will expand its use of multi-component assays because companies want to simplify their quality checks and save money on tests. Endotoxin testing kit sales will grow faster when manufacturers use multi-component equipment in their high-volume production facilities. Companies that create and sell advanced testing solutions which test multiple aspects at once will gain market dominance because customers need better quality control tools.
- Growing Demand for High-Sensitivity Assays: The biopharmaceutical industry requires more sensitive endotoxin testing due to rising product complexity especially with biologics. To keep patients safe from endotoxin risk Complex biopharmaceuticals like monoclonal antibodies and gene therapies need precise endotoxin detection methods. The biopharmaceutical sector needs advanced endotoxin testing kits that precisely identify small endotoxin amounts in intricate sample types as the field grows. The market demand for advanced endotoxin assay kits will grow because they need to detect endotoxins better. Market predictions show endotoxin testing kit demand will increase because drug development becomes more complex while biologics makers focus on product safety. Endotoxin assay technology will gain increased market demand from specialized drug development sectors like biologics and cell therapy fields. Companies that make advanced endotoxin testing kits will grow their market presence because customers want better accuracy in their testing.
Endotoxin Assay Kits Market Opportunities
- Increasing Demand for Point-of-Care Testing Solutions: Point-of-care testing solutions create a strong growth area for the Endotoxin Assay Kits Market. People are using POC testing more often because it provides fast results outside of standard lab facilities. Doctors and nurses could test for endotoxins sooner with on-site detection which would improve emergency care and vaccine manufacturing. POC testing systems help monitor endotoxin contamination in medical products by showing results in real-time so staff can act quickly. The rising market need for POC diagnostic tools will create demand for endotoxin assay kits that are simple to operate and deliver fast results in portable designs. According to market projections healthcare organizations and regulatory authorities will support POC technology adoption which will boost sales of POC endotoxin assay kits. Manufacturers who can design and develop innovative, user-friendly endotoxin testing solutions for POC applications will have the opportunity to capture a larger market share as this trend continues to gain momentum globally.
- Expansion of Biologics and Vaccine Production: The Endotoxin Assay Kits Market can grow through increased vaccine and biologics development. Companies worldwide now prioritize testing methods to keep complex pharmaceutical products safe because biologics and vaccines dominate modern medicine development. Endotoxins trigger dangerous patient responses in many biologic treatments especially monoclonal antibodies cell therapies and gene therapies. Quality control and production of biologics depend on reliable methods to test for endotoxins. Endotoxin testing has taken center stage as vaccine production rates increased during the COVID-19 pandemic. When vaccine producers expand output to serve worldwide needs they must test their formulations for endotoxin content at safe levels. The rising demand for biologics and vaccines creates an excellent chance for endotoxin assay kit manufacturers to develop advanced testing systems. Business projections show that endotoxin testing demand will increase at the same rate as the biologics and vaccine industries develop. Specialized endotoxin assay kit companies will take over more market share because industries making drugs and vaccines require better testing systems every day.
- Technological Advancements in Endotoxin Testing: The Endotoxin Assay Kits Market will grow through new technological developments in testing methods. The Limulus Amebocyte Lysate (LAL) assay remains the primary testing method for endotoxins though it has been used for many years. New biotechnology methods help find endotoxins better and faster while saving money and being more precise. The recombinant factor C (rFC) testing method yields better consistency and less variation compared to previous endotoxin detection methods because it uses synthetic proteins instead of animal products. The latest biosensors detect endotoxin in real time and can operate inside automated systems for large-scale testing. The endotoxin assay kit market will grow because new technology adoption helps pharmaceutical businesses and research organizations measure endotoxins faster and better. The market size of endotoxin testing kits will grow because customers want better automated testing technology. Manufacturers who invest in advanced technology to create top-quality endotoxin tests will establish themselves as market leaders compared to their competitors.
Endotoxin Assay Kits Market Regional Insights
The regional trends and factors influencing the Endotoxin Assay Kits Market throughout the forecast period have been thoroughly explained by the analysts at Insight Partners. This section also discusses Endotoxin Assay Kits Market segments and geography across North America, Europe, Asia Pacific, Middle East and Africa, and South and Central America.
- Get the Regional Specific Data for Endotoxin Assay Kits Market
Endotoxin Assay Kits Market Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2024 | US$ XX million |
| Market Size by 2031 | US$ XX Million |
| Global CAGR (2025 - 2031) | 12.3% |
| Historical Data | 2021-2023 |
| Forecast period | 2025-2031 |
| Segments Covered |
By Testing Method
|
| Regions and Countries Covered | North America
|
| Market leaders and key company profiles |
Endotoxin Assay Kits Market Players Density: Understanding Its Impact on Business Dynamics
The Endotoxin Assay Kits Market market is growing rapidly, driven by increasing end-user demand due to factors such as evolving consumer preferences, technological advancements, and greater awareness of the product's benefits. As demand rises, businesses are expanding their offerings, innovating to meet consumer needs, and capitalizing on emerging trends, which further fuels market growth.
Market players density refers to the distribution of firms or companies operating within a particular market or industry. It indicates how many competitors (market players) are present in a given market space relative to its size or total market value.
Major Companies operating in the Endotoxin Assay Kits Market are:
- Accugen Labs, Wako Chemicals USA, Inc.
- Charles River Laboratories, Inc.,
- Pacific BioLabs
- Sigma-Aldrich Co. LLC.
- Lonza
- STERIS,
Disclaimer: The companies listed above are not ranked in any particular order.
- Get the Endotoxin Assay Kits Market top key players overview
Key Selling Points
- Comprehensive Coverage: The report comprehensively covers the analysis of products, services, types, and end users of the Endotoxin Assay Kits Market, providing a holistic landscape.
- Expert Analysis: The report is compiled based on the in-depth understanding of industry experts and analysts.
- Up-to-date Information: The report assures business relevance due to its coverage of recent information and data trends.
- Customization Options: This report can be customized to cater to specific client requirements and suit the business strategies aptly.
The research report on the Endotoxin Assay Kits Market can, therefore, help spearhead the trail of decoding and understanding the industry scenario and growth prospects. Although there can be a few valid concerns, the overall benefits of this report tend to outweigh the disadvantages.
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Global, Regional, Country
- Industry and Competitive Landscape
- Excel Dataset

Report Coverage
Revenue forecast, Company Analysis, Industry landscape, Growth factors, and Trends
Segment Covered
This text is related
to segments covered.

Regional Scope
North America, Europe, Asia Pacific, Middle East & Africa, South & Central America
Country Scope
This text is related
to country scope.
Frequently Asked Questions
The Endotoxin Assay Kits Market is estimated to witness a CAGR of 12.3% from 2025 to 2031
The major factors driving the Endotoxin Assay Kits Market are Increasing Demand for Pharmaceutical and Biopharmaceutical Products, Growing Stringent Regulatory Requirements, and Advancements in Endotoxin Testing Technology.
Future trends in the Endotoxin Assay Kits Market are Increasing Shift Toward Automated Testing Systems, Focus on Multi-Component Assays, and Growing Demand for High-Sensitivity Assays.
Some of the players operating in the market are Accugen Labs, Wako Chemicals USA, Inc., Charles River Laboratories, Inc., Pacific BioLabs, Sigma-Aldrich Co. LLC., Lonza, STERIS, Nelson Laboratories, LLC, Bio-Synthesis Inc, and Biogenuix.
The report can be delivered in PDF/PPT format; we can also share an excel datasheet based on the request.
Some customization options available based on the request are an additional 3–5 company profiles and a country-specific analysis of 3–5 countries of your choice. Customizations are to be requested/discussed before making final order confirmation# as our team would review the same and check the feasibility.
Trends and growth analysis reports related to Life Sciences : READ MORE..
1. Accugen Labs
2. Wako Chemicals USA, Inc.
3. Charles River Laboratories, Inc.
4. Pacific BioLabs
5. Sigma-Aldrich Co. LLC.
6. Lonza
7. STERIS
8. Nelson Laboratories, LLC
9. Bio-Synthesis Inc
10. Biogenuix
 Get Free Sample For
Get Free Sample For